
Our Services

Human Factor Studies
- Provide clinical assessment of devices including designing and running medical device studies.
- It is so important to thoroughly explore how effective a new healthcare device is going to be and therefore C-TRIC can facilitate focus group to get un-biased genuine feedback from targeted populations.

Project Management
- Carefully assess specific client needs to provide comprehensive protocol delivery.
- Promptly execute our parts in the study.
- Build a strong relationship with client with regular contact.
- Successful completion and time considerations.

Participant Recruitment
- Northern Ireland wide and cross border capabilities to include primary care, secondary care and community recruitment methods.
- We understand the importance of generating patient focused material which is clear and easy to understand and suitable for ethics.
- Based at Altnagelvin Area Hospital with access to NHS patients for recruitment.
- Patient recruitment and sample collection for studies by providing access to skilled research nurses who can help with recruitment, sample collection and label clinical materials, the collation of clinical data and phlebotomy services.

Ethics and Governance Applications
- Accurate and thorough completions of the Integrated Research Application System form (IRAS).
- We handle the process of getting approval without unnecessary delays and resubmission.
- Research Ethics and Governance advice from our in-house clinical governance staff that can help steer you through the process of acquiring governance and ethical approval.
- Staff have an expansive knowledge from previous successful submissions and approvals.
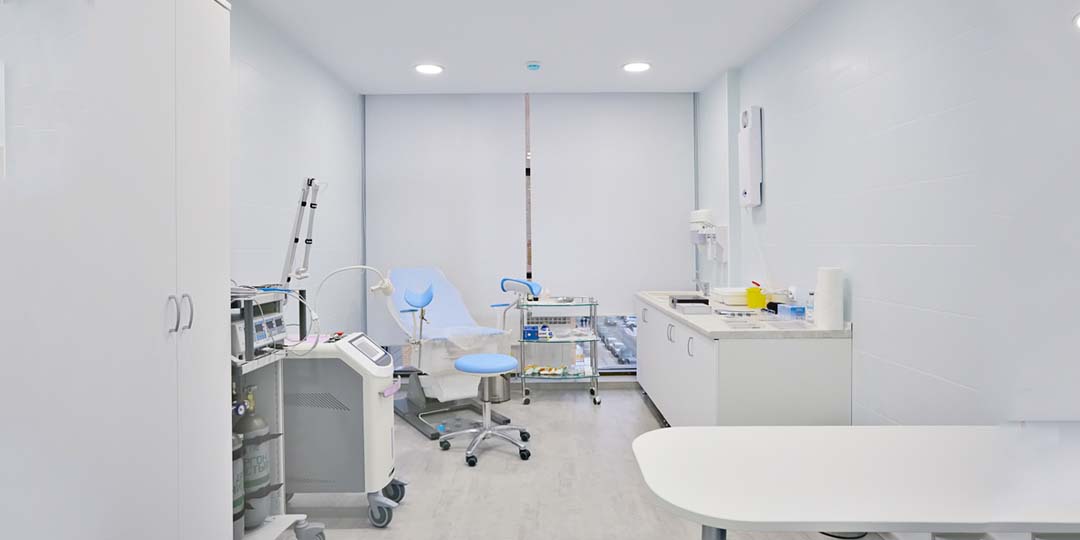
Clinical room availability for professional sample collection
- We understand the importance of providing a safe and trusting environment to our patients/clients. The decision to take part in research can be daunting for many age ranges and those with specific disease areas. By having fully operational clinical rooms we ensure each patient/client’s privacy and dignity throughout the recruitment process.
- All personnel are GCP trained.

Analytical and comparative studies for medical devices
- Bringing a device to market can be costly and therefore as part of our services we can carry out comparative and analytical studies to test your device and compare it to existing devices.
- Reducing overall costs by identifying potential issues at an earlier stage.
- Access to skilled biomedical scientists to process and store clinical materials.

Laboratory access for a range of specialist equipment
- With our fully equipped laboratories we can provide specialist equipment for your project.

Sample storage (HTA licensed facility)
- We have an HTA regulated site with all members fully trained.
- We regularly interact with HTA and run in-house training sessions.

Access to NHS consultants, doctors, research nurses and allied health professionals
- Based in the Western Trust we work alongside our consultants to obtain the desired population for your study.
- Through a multi- disciplinary approach, the work is carried out promptly preventing any delays to your study time scales.
- We can source CIs and PIs for your studies.

In vitro / In vivo specialist work to include proof of concepts / pilot studies
- With in-house facilities and HTA regulated storage facilities we can perform the initial tests, generating your primary results.
- Access to specialist staff who are experts across their fields.

